Congratulations to Dr. Krista Meserve on her recent first author publication in PLOS One. Congratulations also to Dr. Cole Chapman, Dr. Heather Robison, and 3rd-year PhD Candidate Manik Reddy for their contributions to this work.
Multiplexed cytokine profiling identifies diagnostic signatures for latent tuberculosis and reactivation risk stratification
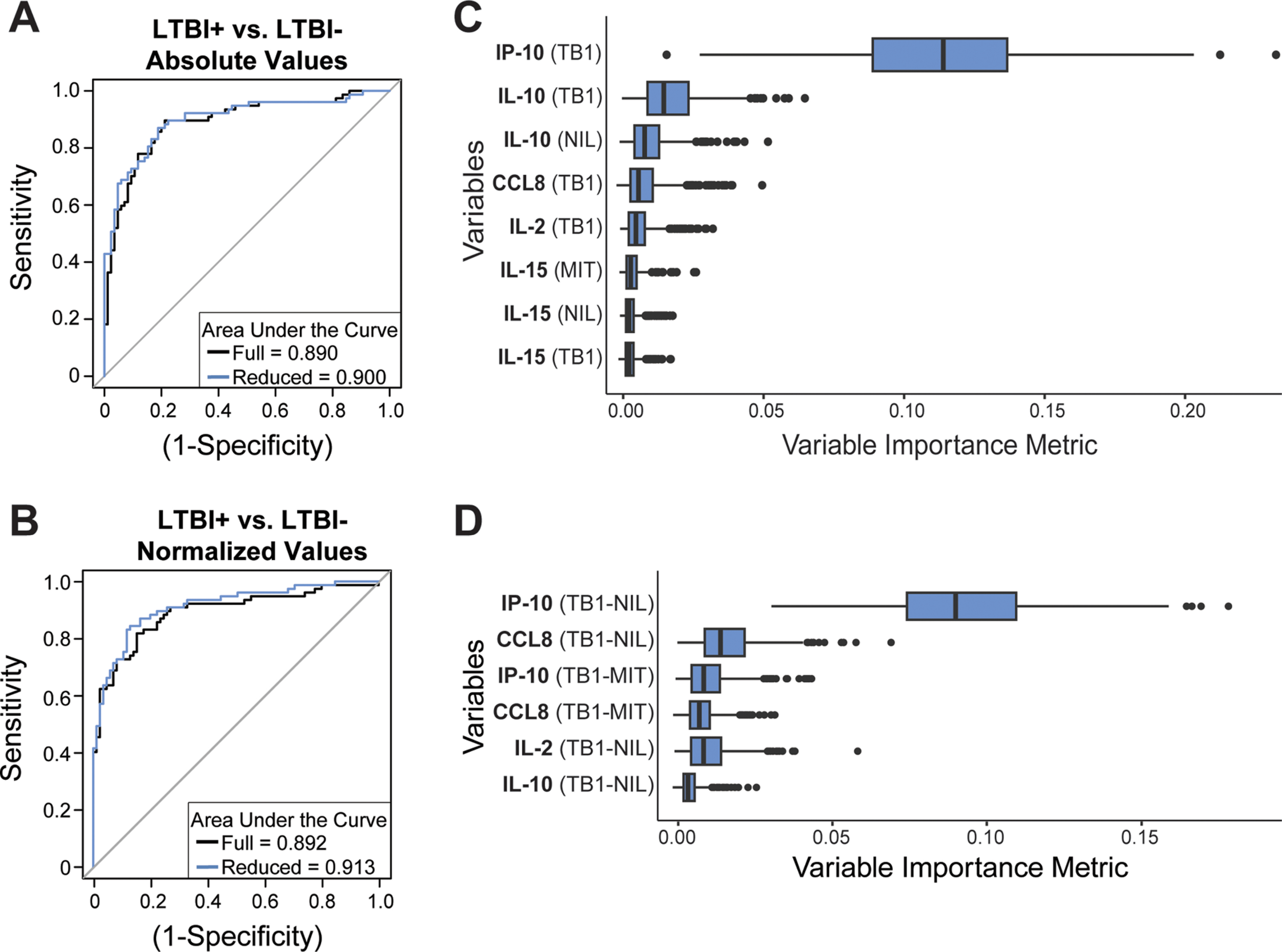
Abstract: Active tuberculosis (TB) is caused by Mycobacterium tuberculosis (Mtb) bacteria and is characterized by multiple phases of infection, leading to difficulty in diagnosing and treating infected individuals. Patients with latent tuberculosis infection (LTBI) can reactivate to the active phase of infection following perturbation of the dynamic bacterial and immunological equilibrium, which can potentially lead to further Mtb transmission. However, current diagnostics often lack specificity for LTBI and do not inform on TB reactivation risk. We hypothesized that immune profiling readily available QuantiFERON-TB Gold Plus (QFT) plasma supernatant samples could improve LTBI diagnostics and infer risk of TB reactivation. We applied a whispering gallery mode, silicon photonic microring resonator biosensor platform to simultaneously quantify thirteen host proteins in QFT-stimulated plasma samples. Using machine learning algorithms, the biomarker concentrations were used to classify patients into relevant clinical bins for LTBI diagnosis or TB reactivation risk based on clinical evaluation at the time of sample collection. We report accuracies of over 90% for stratifying LTBI + from LTBI– patients and accuracies reaching over 80% for classifying LTBI + patients as being at high or low risk of reactivation. Our results suggest a strong reliance on a subset of biomarkers from the multiplexed assay, specifically IP-10 for LTBI classification and IL-10 and IL-2 for TB reactivation risk assessment. Taken together, this work introduces a 45-minute, multiplexed biomarker assay into the current TB diagnostic workflow and provides a single method capable of classifying patients by LTBI status and TB reactivation risk, which has the potential to improve diagnostic evaluations, personalize treatment and management plans, and optimize targeted preventive strategies in Mtb infections.