Dr. Heather Robison recently successfully defended her thesis titled “Multiplexed Immunoassay Development for Precision Medicine Diagnostics and Protein Characterization using Silicon Photonic Microring Resonators".
Congrats to Heather on her recent publication!
A Guide to Quantitative Biomarker Assay
Development using Whispering Gallery
Mode Biosensors
ABSTRACT
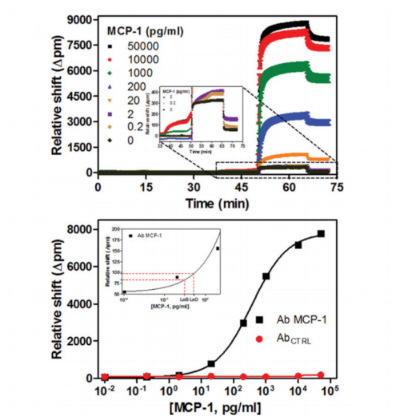
Whispering gallery mode (WGM) sensors are a class of powerful analytical techniques defined by the measurement of changes in the local refractive index at or near the sensor surface. When functionalized with target-specific capture agents, analyte binding can be measured with very low limits of detection. There are many geometric manifestations of WGM sensors, with chip-integrated silicon photonic devices first commercialized because of the robust, wafer-scale device fabrication, facile optical interrogation, and amenability to the creation of multiplexed sensor arrays. Using these arrays, a number of biomolecular targets have been detected in both label-free and label-enhanced assay formats. For example, sub-picomolar detection limits for multiple cytokines were achieved using an enzymatically enhanced sandwich immunoassay that showed high analyte specificity suitable for detection in complex, clinical matrices. This protocol describes a generalizable approach for the development of quantitative, multiplexed immunoassays using silicon photonic microrings as an example WGM platform.
Congrats to Ellen and Sara on the recent publication!
Multiplexed silicon photonic sensor arrays enable facile characterization of coagulation protein binding to Nanodiscs with variable lipid content
ABSTRACT
Interactions of soluble proteins with the cell membrane are critical within the blood coagulation cascade. Of particular interest are the interactions of γ-carboxyglutamic acid-rich (GLA) domain-containing clotting proteins with lipids. Variability among conventional analytical methods presents challenges for comparing clotting protein-lipid interactions. Most previous studies have investigated only a single clotting protein and lipid composition and have yielded widely different binding constants. Herein, we demonstrate that a combination of lipid bilayer Nanodiscs and a multiplexed silicon photonic analysis technology enables high-throughput probing of many protein-lipid interactions among blood-clotting proteins. This approach allowed direct comparison of the binding constants of prothrombin, factor X, activated factor VII, and activated protein C to seven different binary lipid compositions. In a single experiment, the binding constants of one protein interacting with all lipid compositions were simultaneously determined. A simple surface regeneration then facilitated similar binding measurements for three other coagulation proteins. As expected, our results indicated that all proteins exhibit tighter binding (lower Kd) as the proportion of anionic lipid increases. Interestingly, at high proportions of phosphatidylserine, the Kd values of all four proteins began to converge. We also found that although koff values for all four proteins followed trends similar to those observed for the Kd values, the variation among the proteins was much lower, indicating that much of the variation came from the kinetic binding (kon) of the proteins. These findings indicate that the combination of silicon photonic microring resonator arrays and Nanodiscs enables rapid interrogation of biomolecular binding interactions at model cell membrane interfaces.
3rd Annual Bailey Lab Kayaking Trip
The Bailey Lab recently got out into nature on their first kayaking trip since moving to the Wolverine State. While some suffered flip-flop and sunglasses casualties to the Huron River, Ryan managed to stay afloat this time.






Congrats James, Colleen and Josh on the recent publication!
Microfluidic Platform for Efficient Nanodisc Assembly, Membrane Protein Incorporation, and Purification
ABSTRACT
The characterization of integral membrane proteins presents numerous analytical challenges on account of their poor activity under non-native conditions, limited solubility in aqueous solutions, and low expression in most cell culture systems. Nanodiscs are synthetic model membrane constructs that offer many advantages for studying membrane protein function by offering a native-like phospholipid bilayer environment. The successful incorporation of membrane proteins within Nanodiscs requires experimental optimization of conditions. Standard protocols for Nanodisc formation can require large amounts of time and input material, limiting the facile screening of formation conditions. Capitalizing on the miniaturization and efficient mass transport inherent to microfluidics, we have developed a microfluidic platform for efficient Nanodisc assembly and purification, and demonstrated the ability to incorporate functional membrane proteins into the resulting Nanodiscs. In addition to working with reduced sample volumes, this platform simplifies membrane protein incorporation from a multi-stage protocol requiring several hours or days into a single platform that outputs purified Nanodiscs in less than one hour. To demonstrate the utility of this platform, we incorporated Cytochrome P450 into Nanodiscs of variable size and lipid composition, and present spectroscopic evidence for the functional active site of the membrane protein. This platform is a promising new tool for membrane protein biology and biochemistry that enables tremendous versatility for optimizing the incorporation of membrane proteins using microfluidic gradients to screen across diverse formation conditions.
Grill and Chill 2017
Congrats to Alex for defending her thesis!
Congratulations to Dr. Alex Stanton, who today successfully defended her thesis "Organically-modified Silicon Photonic Microring Resonators for Chemical Sensing"!
Congrats to Maria for passing her candidacy exam!
Congrats to Dr. Graybill and Dr. Wade!
They each successfully defended their theses, though the cake may have suggested a different celebration. Congratulations to Dr. Graybill for successfully defending his thesis "Improving Multiplexed RNA Detection Assays By Interfacing Enzymatic Amplification Strategies with Silicon Photonic Microring Resonators.” Congratulations to Dr. Wade, as well. He defended his thesis "Expanding the Toolbox for Personalized Medicine with Silicon Photonic Microring Resonators and Microfluidic Technologies."
Congrats to Steve on his recent publication!
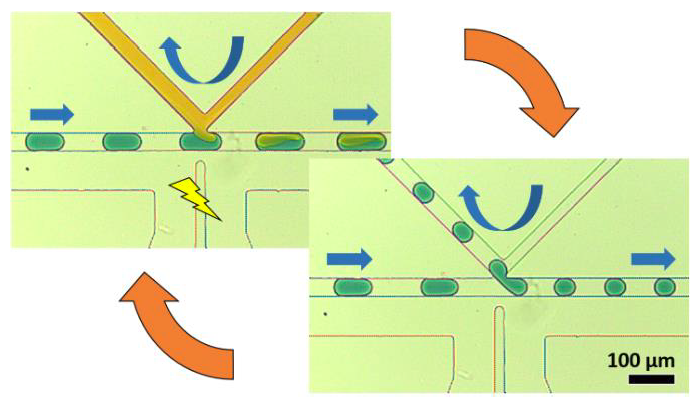
The K-Channel: A Multi-Functional Architecture for Dynamically Re-Configurable Sample Processing in Droplet Microfluidics
ABSTRACT
By rapidly creating libraries of thousands of unique, miniaturized reactors, droplet microfluidics provides a powerful method for automating high-throughput chemical analysis. In order to engineer in-droplet assays, microfluidic devices must add reagents into droplets, remove fluid from droplets, and perform other necessary operations, each typically provided by a unique, specialized geometry. Unfortunately, modifying device performance or changing operations usually requires re-engineering the device among these specialized geometries, a time-consuming and costly process when optimizing in-droplet assays. To address this challenge in implementing droplet chemistry, we have developed the “K-channel,” which couples a cross-channel flow to the segmented droplet flow to enable a range of operations on passing droplets. K-channels perform reagent injection (0-100% of droplet volume), fluid extraction (0-50% of droplet volume), and droplet splitting (1:1-1:5 daughter droplet ratio). Instead of modifying device dimensions or channel configuration, adjusting external conditions, such as applied pressure and electric field, selects the Kchannel process and tunes its magnitude. Finally, interfacing a device-embedded magnet allows selective capture of 96% of dropletencapsulated superparamagnetic beads during 1:1 droplet splitting events at ~400 Hz. Addition of a second K-channel for injection (after the droplet splitting K-channel) enables integrated washing of magnetic beads within rapidly-moving droplets. Ultimately, the K-channel provides an exciting opportunity to perform many useful droplet operations across a range of magnitudes without requiring architectural modifications. Therefore, we envision the K-channel as a versatile, easy to use microfluidic component enabling diverse, in-droplet (bio)chemical manipulations.